ವಿಕಿಪೀಡಿಯದಿಂದ, ಇದು ಮುಕ್ತ ಹಾಗೂ ಸ್ವತಂತ್ರ ವಿಶ್ವಕೋಶ
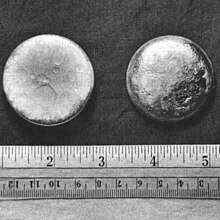
ಪ್ಲುಟೋನಿಯ್, 94 Name, symbol ಪ್ಲುಟೋನಿಯ್, Pu Pronunciation / / ploo-TOH Appearance silvery white, tarnishing to dark gray in air
Atomic number 94 Standard atomic weight Group , block n/a , f-block Period period 7 Electron configuration [Rn ] 5f6 7s2 per shell 2, 8, 18, 32, 24, 8, 2 Phase solid Melting point 912.5 K (639.4 °C, 1182.9 °F) Boiling point 3505 K (3228 °C, 5842 °F) Density (near r.t. ) 19.816 g· cm−3 (at 0 °C, 101.325 kPa ) Liquid density at m.p. : 16.63 g· cm−3 Heat of fusion 2.82 kJ· mol−1 Heat of 333.5 kJ· mol−1 Molar heat capacity 35.5 J· mol−1 · K−1 pressure ಟೆಂಪ್ಲೇಟು:Infobox element/vapor pressure table Oxidation states 8, 7, 6, 5, 4 , 3, 2, 1 (an amphoteric oxide)Electronegativity 1.28 (Pauling scale) energies 1st: 584.7 kJ· mol−1 Atomic radius empirical: 159 pm Covalent radius 187±1 pm Crystal structure monoclinic Speed of sound 2260 m· s−1 Thermal expansion 46.7 µm· m−1 · K−1 Thermal conductivity 6.74 W· m−1 · K−1 Electrical resistivity at 0 °C: 1.460 µΩ· m Magnetic ordering paramagnetic Young's modulus 96 GPa Shear modulus 43 GPa Poisson ratio 0.21 CAS number 7440-07-5 Naming after dwarf planet Pluto , itself named after classical god of the underworld Pluto Discovery Glenn T. Seaborg , Arthur Wahl , Joseph W. Kennedy , Edwin McMillan (1940–1) Main article: Isotopes of ಪ್ಲುಟೋನಿಯ್
· references
ಟೆಂಪ್ಲೇಟು:Infobox element/element navigation
References These references will appear in the article, but this list appears only on this page.
Infoboxes for the chemical elements use the core template {{Infobox element }}.
In general Usually parameters reproduce the value, and the template adds the SI unit or additional standard text. While the topic is technical, we can strive to make the result readable text, and even verbose. Not every reader is the Seaborg you are. For example, this is why the template writes "(at 0 °C)". For this, you may want to experiment with parameter input too - or propose improvements.
All this can vary across the values (data rows). So depending on the row and the specific value, additional formattings may be added by the template (e.g., a newline <br>)
Parameter naming patterns (the _ underscore is a simple space)
..._2, ..._3Numbered values make a list.
..._refMost parameters have a _ref brother that allows for the reference. This reference is added right after the unit.
..._commentParameters with a _comment suffix are added to the end of a line, with a space added. Comments are reproduced without additions (no brackets, no italics, ...).
Parameter list
This parameter list:
{{Infobox element
<!-- top -->
|image name=
|image alt=
|image size=
|image name comment=
|image name 2=
|image alt 2=
|image size 2=
|image name 2 comment=
<!-- General properties -->
|name=
|symbol=
|pronounce=
|pronounce ref=
|pronounce comment=
|pronounce 2=
|alt name=
|alt names=
|allotropes=
|appearance=
<!-- Periodic table -->
|above=
|below=
|left=
|right=
|number=
|atomic mass=
|atomic mass 2=
|atomic mass ref=
|atomic mass comment=
|series=
|series ref=
|series comment=
|series color=
|group=
|group ref=
|group comment=
|period=
|period ref=
|period comment=
|block=
|block ref=
|block comment=
|electron configuration=
|electron configuration ref=
|electron configuration comment=
|electrons per shell=
|electrons per shell ref=
|electrons per shell comment=
<!-- Physical properties -->
|physical properties comment=
|color=
|phase=
|phase ref=
|phase comment=
|melting point K=
|melting point C=
|melting point F=
|melting point ref=
|melting point comment=
|boiling point K=
|boiling point C=
|boiling point F=
|boiling point ref=
|boiling point comment=
|sublimation point K=
|sublimation point C=
|sublimation point F=
|sublimation point ref=
|sublimation point comment=
|density gplstp=
|density gplstp ref=
|density gplstp comment=
|density gpcm3nrt=
|density gpcm3nrt ref=
|density gpcm3nrt comment=
|density gpcm3nrt 2=
|density gpcm3nrt 2 ref=
|density gpcm3nrt 2 comment=
|density gpcm3nrt 3=
|density gpcm3nrt 3 ref=
|density gpcm3nrt 3 comment=
|density gpcm3mp=
|density gpcm3mp ref=
|density gpcm3mp comment=
|density gpcm3bp=
|density gpcm3bp ref=
|density gpcm3bp comment=
|triple point K=
|triple point kPa=
|triple point ref=
|triple point comment=
|triple point K 2=
|triple point kPa 2=
|triple point 2 ref=
|triple point 2 comment=
|critical point K=
|critical point MPa=
|critical point ref=
|critical point comment=
|heat fusion=
|heat fusion ref=
|heat fusion comment=
|heat fusion 2=
|heat fusion 2 ref=
|heat fusion 2 comment=
|heat vaporization=
|heat vaporization ref=
|heat vaporization comment=
|heat capacity=
|heat capacity ref=
|heat capacity comment=
|heat capacity 2=
|heat capacity 2 ref=
|heat capacity 2 comment=
|vapor pressure 1=
|vapor pressure 10=
|vapor pressure 100=
|vapor pressure 1 k=
|vapor pressure 10 k=
|vapor pressure 100 k=
|vapor pressure ref=
|vapor pressure comment=
|vapor pressure 1 2=
|vapor pressure 10 2=
|vapor pressure 100 2=
|vapor pressure 1 k 2=
|vapor pressure 10 k 2=
|vapor pressure 100 k 2=
|vapor pressure 2 ref=
|vapor pressure 2 comment=
<!-- Atomic properties -->
|atomic properties comment=
|oxidation states=
|oxidation states ref=
|oxidation states comment=
|electronegativity=
|electronegativity ref=
|electronegativity comment=
|ionization energy 1=
|ionization energy 1 ref=
|ionization energy 1 comment=
|ionization energy 2=
|ionization energy 2 ref=
|ionization energy 2 comment=
|ionization energy 3=
|ionization energy 3 ref=
|ionization energy 3 comment=
|number of ionization energies=
|ionization energy ref=
|ionization energy comment=
|atomic radius=
|atomic radius ref=
|atomic radius comment=
|atomic radius calculated=
|atomic radius calculated ref=
|atomic radius calculated comment=
|covalent radius=
|covalent radius ref=
|covalent radius comment=
|Van der Waals radius=
|Van der Waals radius ref=
|Van der Waals radius comment=
<!-- Miscellanea -->
|crystal structure=
|crystal structure prefix=
|crystal structure ref=
|crystal structure comment=
|crystal structure 2=
|crystal structure 2 prefix=
|crystal structure 2 ref=
|crystal structure 2 comment=
|speed of sound=
|speed of sound ref=
|speed of sound comment=
|speed of sound rod at 20=
|speed of sound rod at 20 ref=
|speed of sound rod at 20 comment=
|speed of sound rod at r.t.=
|speed of sound rod at r.t. ref=
|speed of sound rod at r.t. comment=
|thermal expansion=
|thermal expansion ref=
|thermal expansion comment=
|thermal expansion at 25=
|thermal expansion at 25 ref=
|thermal expansion at 25 comment=
|thermal conductivity=
|thermal conductivity ref=
|thermal conductivity comment=
|thermal conductivity 2=
|thermal conductivity 2 ref=
|thermal conductivity 2 comment=
|thermal diffusivity=
|thermal diffusivity ref=
|thermal diffusivity comment=
|electrical resistivity=
|electrical resistivity unit prefix=
|electrical resistivity ref=
|electrical resistivity comment=
|electrical resistivity at 0=
|electrical resistivity at 0 ref=
|electrical resistivity at 0 comment=
|electrical resistivity at 20=
|electrical resistivity at 20 ref=
|electrical resistivity at 20 comment=
|band gap=
|band gap ref=
|band gap comment=
|Curie point K=
|Curie point ref=
|Curie point comment=
|magnetic ordering=
|magnetic ordering ref=
|magnetic ordering comment=
|tensile strength=
|tensile strength ref=
|tensile strength comment=
|Young's modulus=
|Young's modulus ref=
|Young's modulus comment=
|Shear modulus=
|Shear modulus ref=
|Shear modulus comment=
|Bulk modulus=
|Bulk modulus ref=
|Bulk modulus comment=
|Poisson ratio=
|Poisson ratio ref=
|Poisson ratio comment=
|Mohs hardness=
|Mohs hardness ref=
|Mohs hardness comment=
|Mohs hardness 2=
|Mohs hardness 2 ref=
|Mohs hardness 2 comment=
|Vickers hardness=
|Vickers hardness ref=
|Vickers hardness comment=
|Brinell hardness=
|Brinell hardness ref=
|Brinell hardness comment=
|CAS number=
|CAS number ref=
|CAS number comment=
<!-- History -->
|naming=
|predicted by=
|prediction date ref=
|prediction date=
|discovered by=
|discovery date ref=
|discovery date=
|first isolation by=
|first isolation date ref=
|first isolation date=
|named by=
|named date ref=
|named date=
|history comment label=
|history comment=
<!-- Isotopes -->
|isotopes=
|isotopes comment=
|engvar=
}}
{{Infobox element }}; labels & notes:
TOP
(Image)
(Image 2)
GENERAL PROPERTIES
Name
symbol
Pronunciation
Alternative name(s)
Allotropes
Appearance
<element> IN THE PERIODIC TABLE
Periodic table
Atomic number
Standard atomic weight
()
Element category
(set headers background color)
Group
Period
Block
Electron configuration
Electrons per shell
PHYSICAL PROPERTIES
(general note)
Color
Phase
Melting point
Boiling point
Sublimation point
Density
Density
Liquid density
Liquid density
Triple point
Critical point
Heat of fusion
Heat of vaporization
Molar heat capacity
Vapor pressure
Vapor pressure
ATOMIC PROPERTIES
Oxidation states
Electronegativity
Ionization energy
Atomic radius
Covalent radius
Van der Waals radius
MISCELLANEA
Crystal structure
Crystal structure
Speed of sound
Thermal expansion
Thermal conductivity
Thermal diffusivity
Electrical resistivity
Band gap
Curie point
Magnetic ordering
Tensile strength
Young's modulus
Shear modulus
Bulk modulus
Poisson ratio
Mohs hardness
Vickers hardness
Brinell hardness
CAS Number
HISTORY
Naming
Prediction
()
Discovery
()
First isolation
()
Named by
()
(History comment)
ISOTOPES OF <element>
Isotopes of <element>
(wp:engvar )
Isotopes
Isotopes are to be entered by the editor using these subtemplates:
Examples (from various elements):
| isotopes =
{{Infobox element/isotopes stable
| link=Fluorine-19 | mn=19 | sym=F | na=100% | n=10 }}
{{Infobox element/isotopes decay
| mn=251 | sym=Cf
| na=trace | hl=898 y
| dm=α | de=6.172 | link1=curium-247 | pn=247 | ps=Cm}}
{{Infobox element/isotopes decay2 | mn=252 | sym=Cf
| na=trace | hl=2.645 y
| dm1=α (96.91%)| de1=6.217 | link1=curium-248 | pn1=248 | ps1=Cm
| dm2=SF (3.09%)| de2=– | pn2= | ps2=–}}
{{Infobox element/isotopes decay3
| mn=26 | sym=Al | na=[[trace radioisotope|trace]] | hl={{val|7.17|e=5|ul=y}}
| dm1=[[Positron emission|β<sup>+</sup>]] | de1=1.17 | link1=magnesium-26
| pn1=26 | ps1=Mg
| dm2=[[electron capture|ε]] | de2=- | link2=magnesium-26 | pn2=26 | ps2=Mg
| dm3=[[Gamma radiation|γ]] | de3=1.8086 | pn3= | ps3=-
}}
{{Infobox element/isotopes decay3 (2 2 1)
| mn=22 | sym=Na | na=[[trace radioisotope|trace]]
| hl=[[1 E7 s|2.602]] [[year|y]] | dm1=[[Beta decay|β<sup>+</sup>]]→[[Gamma decay|γ]]
| de1a=0.5454 | link1a=neon-22 | pn1a=22 | ps1a=Ne*
| de1b=1.27453(2)<ref name=npa521_1>{{cite journal
| last=Endt | first=P. M.
| title=Energy levels of A = 21–44 nuclei (VII)
| journal=Nuclear Physics A | volume=521 | pages=1–400
| date=1990 | doi=10.1016/0375-9474(90)90598-G
| bibcode=1990NuPhA.521....1E
}}</ref> | pn1b=22 | ps1b=Ne
| dm2=[[electron capture|ε]]→[[Gamma decay|γ]]
| de2a=- | pn2a=22 | ps2a=Ne*
| de2b=1.27453(2)
| pn2b=22 | ps2b=Ne
| dm3=[[Beta decay|β<sup>+</sup>]] | de3=1.8200
| pn3=22 | ps3=Ne }}
{{Infobox element/isotopes_decay4 | mn=260 | sym=Md
| na=[[synthetic radioisotope|syn]] | hl=31.8 [[day|d]]
| dm1=SF | de1=- | pn1= | ps1=-
| dm2=α | de2=7.000 | link2=einsteinium-256 | pn2=256 | ps2=Es
| dm3=ε | de3=- | link3=fermium-260 | pn3=260 | ps3=Fm
| dm4=[[beta emission|β<sup>−</sup>]]
| de4=1.000 | link4=nobelium-260 | pn4=260 | ps4=No}}
|isotopes comment=reference{{sfn|Chisté|2006}}1310<br>
* = [[excited state]]
produces the subtable:
Other subtemplates are used automatically; they do not need specific editor's input.
{{Periodic table (32 columns, micro) }}When atomic number ≥ 119, the miniature periodic table shows an extended version (period 8 and up). See, for example, {{Infobox ununennium }}. References These references will appear in the article, but this list appears only on this page.
↑ Endt, P. M. (1990). "Energy levels of A = 21–44 nuclei (VII)". Nuclear Physics A . 521 : 1–400. Bibcode :1990NuPhA.521....1E . doi :10.1016/0375-9474(90)90598-G .